Organic Lewis Acid Catalysis
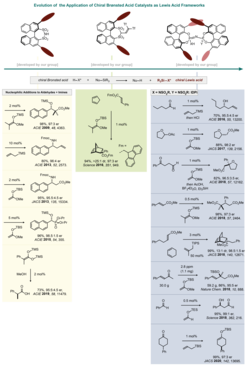
To date, the field of Lewis acid catalysis has been dominated by (transition) metal chemistry. To provide purely organic counterparts to these traditional systems, we are dedicated to the synthesis and identification of organic Lewis acids. In 2009, we demonstrated that our chiral disulfonimides (DSI) are not only excellent Brønsted acids, but also form strong Lewis acids upon silylation. These in situ generated Lewis acids facilitated a variety of enantioselective silyl transfer reactions. Our recently developed, confined and highly acidic imidodiphosphorimidate (IDPi) catalysts have overcome previous limitations and were able to solve long-standing problems in this field, such as the enantioselective Hosomi–Sakurai allylation of aldehydes.
