EPR, Raman and Hydrogen Production
The focus of our group is on the understanding of general processes in catalysis by spectroscopic investigation of the electronic structure of reaction intermediates. Our main research methods are resonance Raman spectroscopy, electron paramagnetic resonance spectroscopy (EPR) and quantum chemistry. The knowledge of the electronic structure of catalytic intermediates is of crucial importance, since electrons play a major role in almost all catalytic processes, which are almost exclusively based on redox chemistry. Whenever electrons are in motion, the knowledge of where the redox-active electrons are located, e.g. in the form of donor and acceptor orbitals, is crucial both for the understanding and for the gradual improvement of the catalysts. In addition, the knowledge gained by combining these methods can also be used to identify transition states and energy barriers that may need to be overcome in order to further improve catalysis.
Research Topics:
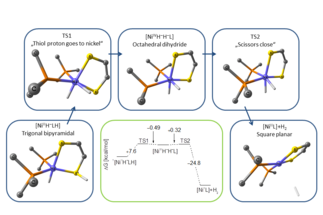
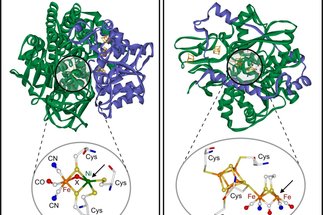
The hydrogen economy represents a way of fulfilling our need for energy by using molecular hydrogen as an energy carrier and using reactions in which polluting products like greenhouse gases are avoided. The prospects of such...
[more]
The hydrogen economy represents a way of fulfilling our need for energy by using molecular hydrogen as an energy carrier and using reactions in which polluting products like greenhouse gases are avoided. The prospects of such an economy require the development of clean and efficient ways of producing and storing molecular hydrogen, or, in an extended sense, of molecules by which energy is stored in their chemical bonds.
[more]
Raman and resonance Raman spectroscopy are well established methods to investigate the vibrational frequencies of molecular systems. In particular, in the area of catalysis, one of the focal points of the MPI für Kohlenforschung, this spectroscopy is a valuable tool for acquiring experimental information about the electronic structure of catalysts and thereby of the catalytic mechanism. This can be achieved by ...
[more]